- Augusta University
- Georgia Cancer Center
- Research
- Shared Resources
- Biorepository
- Biorepository Alliance of Georgia (BRAG-Onc)
Biorepository Alliance of Georgia (BRAG-Onc)
The mission of the Biorepository Alliance of Georgia (BRAG-Onc) is to provide a centralized service for biospecimen procurement and distribution across the State of Georgia to advance cancer research.
BRAG-Onc was established to represent the diversity of the cancer patient population in Georgia and to enhance cancer research in the state. The repository collects and stores specimens under standardized conditions with accompanying clinical and demographic information. The collection is supported by a web-accessible database for inventory management and annotation, and a long-term storage facility with backups of cryopreserved specimens. The repository serves as the central coordinating center for the statewide network.
Contact Us
Biorepository
Project Overview
A few representative studies are described below to demonstrate the effectiveness of the Alliance in supporting basic and translational research. Over the last four years, the Biorepository has supported over 30 individual projects.

In addition to the projects presented below, the biorepository has been involved in The Cancer Genome Atlas (TGCA) project. This very important undertaking is currently the National Cancer Institute’s (NCI’s) flagship research program, involving both intramural NCI scientists as well as extramural scientists from leading research centers and universities around the globe. In 2003, the first complete sequencing of the entire human genome was completed at a cost of over $3-billion. Sequencing technology has since exponentially improved, bringing the cost and time required down to remarkable levels in just a decade. In 2009, the NHGRI and NCI launched the full-scale TCGA program, with the goal to perform in-depth molecular analysis and sequencing of 4,000 cancer patients from the 20 most common cancers.
Since 2010, BRAG-Onc has been a vital member of the TCGA network and has contributed specimens from multiple cancer types including Head and Neck cancer, Thyroid cancer, Ovarian cancer, and Urinary Bladder cancer. Inclusion in the TCGA program earmarks BRAG-Onc as meeting the most stringent quality control standards for specimen processing, alongside peer institutions such as MD Anderson, Fox Chase Cancer Institute, Memorial Sloan-Kettering, Imperial College London, and UCSF.
While it will likely take years to fully analyze the wealth of data being produced, the promises of this effort are already being realized. In addition to the NCI grant to fund the specimen collection and annotation, GRU Cancer Center physicians Weinberger and Bollag were recently included as co-authors in a major TCGA study published in the journal Nature. Further publications and investigator-initiated grant proposals are also anticipated.
Acute Myelogenous Leukemia
John Cowell, PhD, FRCPath is the Director of the Molecular Oncology and Biomarker Program and Associate Director for Basic Research at the Georgia Cancer Center. His lab has been a leader in understanding the progression of leukemic stem cells to acute myelogenous leukemia (AML). Samples obtained from the biorepository have helped understand the molecular etiology of AML in specific genetic subtypes and provides opportunities to develop novel treatment regimens.
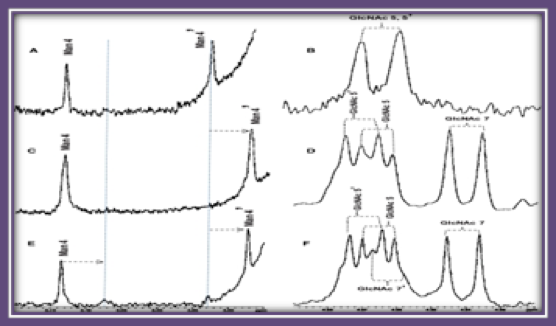
Targeted glycoproteomic identification of biomarkers for human breast carcinoma
Dr. Michael Pierce, Director of the University of Georgia Cancer Center, has also been using tissues from the Biorepository to study the role of glycans in cancer. Research by Pierce and others has shown that changes in cell glycans can herald the presence of cancerous or precancerous cells. Among his research accomplishments, Pierce and his team isolated a specific enzyme known as Gnt-V that is elevated in colorectal and breast cancer cells, as well as other types of cancer. The team is now investigating ways to inhibit Gnt-V in hopes of developing a treatment that will slow the growth of tumors and prevent metastasis.
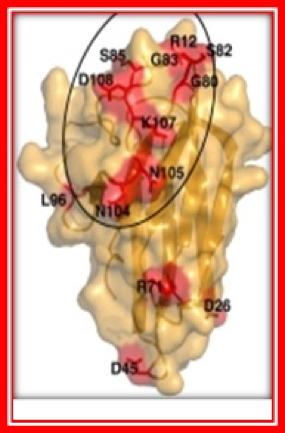
The Role of Indolamine-2,3 dioxygenase (IDO) in the suppression of Anti-tumor Immune Responses
Cells from the tumor bank have also been used by Dr. David Munn, Professor of Pediatrics, who works on tumor immunology and the molecular mechanisms of immune suppression and tolerance. A major focus of the laboratory is the role of tryptophan metabolism by the enzyme indolamine-2, 3 dioxygenase (IDO). IDO-expressing dendritic cells are studied with an emphasis on how these cells suppress anti-tumor immune responses via IDO and downstream pathways. The laboratory employs clinical and translational strategies designed to enhance antitumor immune responses using IDO-inhibitor drugs.

Heat shock Protein 90 as a Therapeutic Target
The focus in the Chadli laboratory is on the identification of heat shock protein 90 (Hsp90) inhibitors as anti-cancer reagents. The Hsp90 chaperoning machine maintains the conformation and stability of many oncogenic proteins, transcription factors, steroid receptors, metalloproteases, and nitric oxide synthases that are essential for cancer cell survival and proliferation. The Hsp90 machine is therefore an exciting therapeutic target, the inactivation of which would deliver a combinatorial attack on multiple signaling pathways, leading to a more efficient killing of cancer cells and reducing resistance to chemotherapy. His laboratory has developed novel small molecule inhibitors of the Hsp90 chaperoning machine and has used samples from the biorepository to test the efficacy of these new drugs.
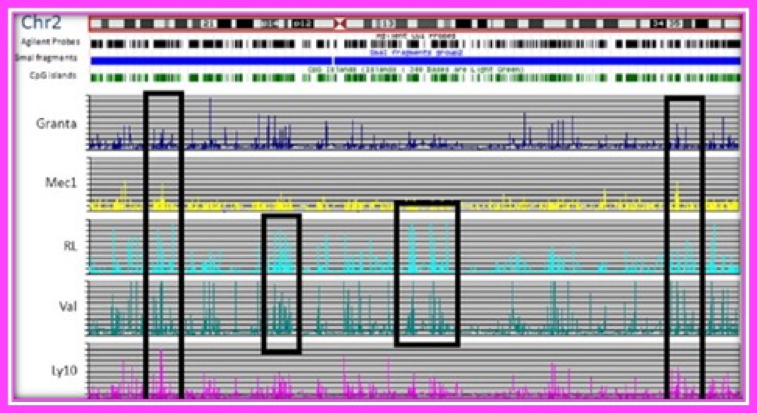
Whole Methylome Sequencing in Lymphoid Malignancies
The Shi laboratory has spent a significant amount of effort on developing high-throughput technologies for dissecting complex epigenetic regulation in normal and cancer cells. One of their most recent innovations was the development of a targeted bisulfite sequencing method based on solution-based sequence capture technology. Currently, they are utilizing these high-throughput bisulfite-sequencing methods for profiling DNA methylation patterns in leukemias and lymphomas. They have demonstrated that aberrant epigenetic gene regulation in lymphoid malignancies involves DNA methylation, histone modifications, and spatial conformation of the genome. Samples obtained through the Biorepository have been used to elucidate the epigenetic profiles in chronic lymphocytic leukemia which can now distinguish subtypes that have better outcomes.
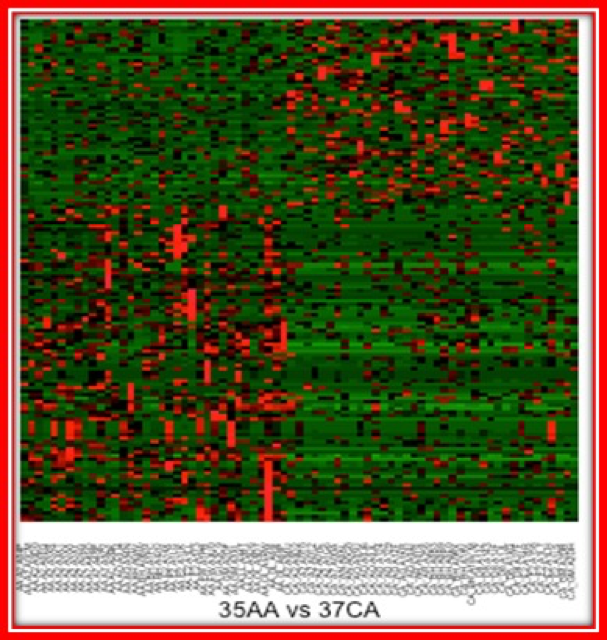
Mutational Analysis of Triple Negative Breast Cancers to Examine Racial Disparities in Outcomes
The Hawthorn Laboratory is in the process of sequencing triple negative breast cancers (TNBCs) obtained from the biorepository. Triple negative breast cancer is defined by the absence of detectable estrogen and progesterone receptor expression levels and by the lack of the epidermal growth factor receptor 2 (HER2) gene amplification. Because it is lacking these tumor-specific molecules there are currently no targeted therapies to treat this type of breast cancer. The survival rate of women developing this breast cancer subtype is a devastating 14% while those who develop estrogen positive tumors have a survival rate of 97%. Women of African descent (AA) have a generally lower rate of breast cancer incidence than women of European descent (CA), however, 20% of tumors in CA patients are TNBCs, while ~40% of breast cancers in AA patients are TNBCs. Dr. Hawthorn is using whole genome sequencing to profile TNBC tumors and examining the profiles of CA and AA patients to determine if the disease is different between the two racial groups. There is currently very limited data on AA TNBC as a separate disease entity so that access to the samples provided by BRAG-Onc are critical to the success of this important study.
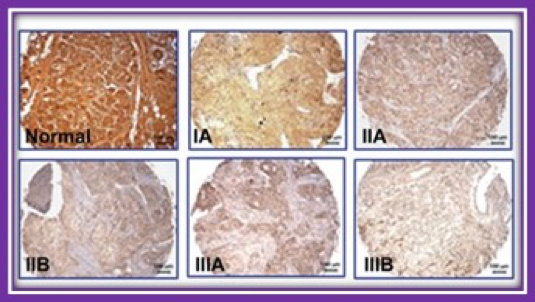
The Niacin/Butyrate Receptor GPR109A Suppresses Mammary Tumorigenesis By Inhibiting Cell Survival
Drs. Ganapathy and Thangaraju of the Signaling and Angiogenesis Program within Georgia Cancer Center have studied the tumor suppressor function of a receptor for niacin and butyrate called GPR109A. Using samples received from BRAG-Onc, these investigators found that GPR109A is expressed in normal breast tissue but is silenced in tumor tissue. Studies have shown that activation of this receptor results in the inhibition of genes involved in cell survival of breast cancer cells. These findings have been confirmed in cell lines and mouse models of breast cancer and suggest the GPR109A is a tumor suppressor in the mammary gland. Studies are now underway to determine if therapeutic activation of this gene may be an effective strategy in the treatment of breast cancer.
BRAG-Onc Membership
Here is a look at the current medical centers, clinics, etc. who are members of the Biorepository Alliance of Georgia (BRAG-Onc).