Core C — Endothelial Cell Analytic Core
Overall Goal of Core C
The overall goal of the Cell and Analytical Core is to facilitate completion of the Specific Aims outlined in the scientific Projects 1-3 by providing well-characterized primary endothelial cells from wild type and transgenic mice and human tissue, measurements of endothelial cell (EC) metabolism, barrier function, EndMT, generation of reactive oxygen and nitrogen species and inflammation.
Central Hypothesis
Excess ROS induced by cardiovascular risk factors such as diabetes can impair the balance between redox signaling and metabolism, resulting in endothelial dysfunction (e.g. impaired angiogenesis), which contributes to vascular disease, such as peripheral artery disease (PAD). Preliminary data support the hypothesis that VEGF/NOXderived H2O2 stimulates Cys oxidation of Drp1 to promote mitochondrial fission and mitoROS, which drives glycolysis and endothelial activation (e.g. angiogenesis) by oxidative activation of AMPK as well as increased PFKFB3 expression in ECs. By contrast, in disease conditions such as diabetes, this pathway is impaired due to hyperactivation of Drp1 via phosphorylation which promotes pathological mitochondrial fragmentation-excess mitoROS that leads to VEGFR2 degradation.
Core Aims:
Aim 1 – Cell isolation & characterization –service component
To uniformly isolate primary endothelial cells from wild-type and transgenic mice,
as well as from Type 1 diabetes patients. Standardized characterization of isolated EC and to
expand and distribute purified, characterized EC in early passages to three research projects.
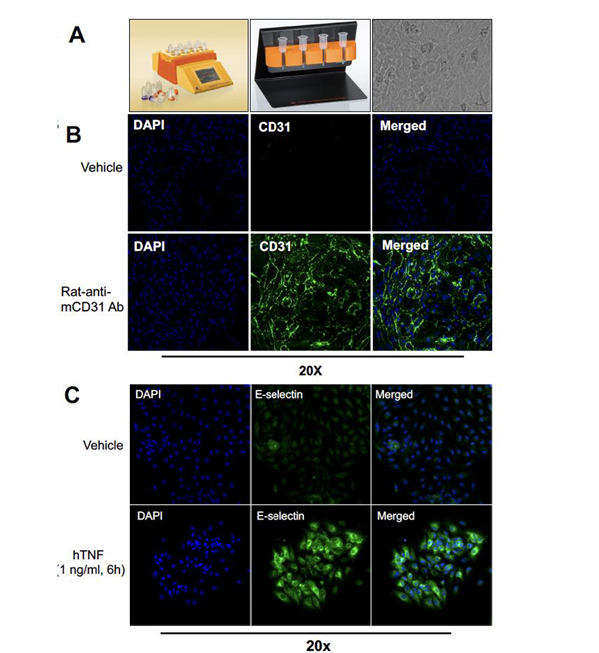
Figure C1 Title: Endothelial Cell isolation and characterization.
- Purification setup for primary mouse endothelial cells using a dissociator and magnetic
bead selection
(CD45−/CD31+), with example lung endothelial cells showing cobblestone morphology. - DAPI-stained nuclei and CD31-labeled endothelial cells in purified mouse lung samples.
- DAPI and E-selectin staining of human lung endothelial cells ± TNF treatment.
Aim 2 – Gene delivery & deletion –service component
To construct, purify, and deliver recombinant adenovirus, lentivirus, and adeno-associated
virus. Adenovirus and adeno-associated virus (AAV). To deplete and manipulate endogenous
genes in mouse and human EC
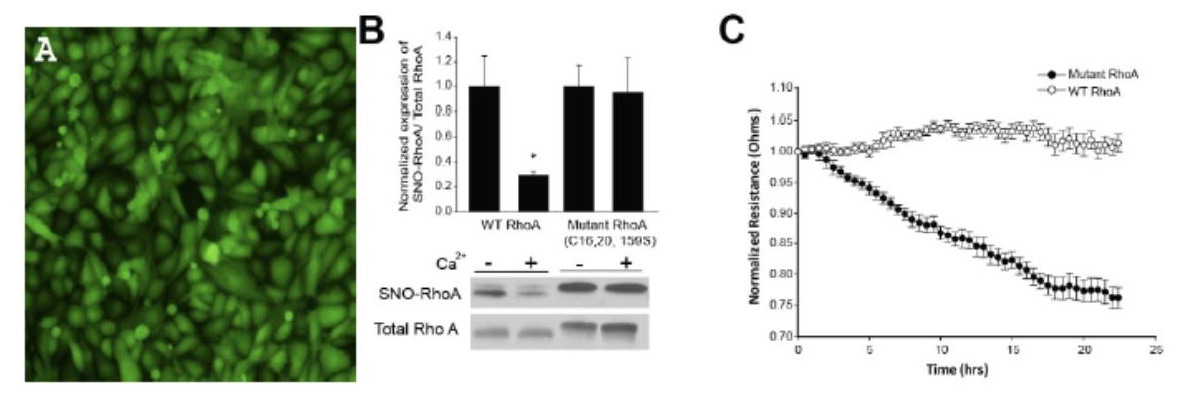
Figure C2 Title: Adenovirus-mediated gene expression in human lung microvascular endothelial cells.
- Green Fluorescent Protein expression.
- HLMVEC were transduced with adenoviruses encoding wild-type or mutant RhoA, lysed and then incubated with or without 3mM CaCl2.
- Normalized Transendothelial resistance of HLMVEC monolayers transduced with wild-type or mutant RhoA viruses.
Aim 3 – Analysis – academic component
Metabolic analysis, to measure reactive oxygen (ROS) and nitrogen species (RNS). Detect
EC inflammation, to access EndMT-related genes in ECs by transcriptomics, to measure
endothelial monolayer permeability
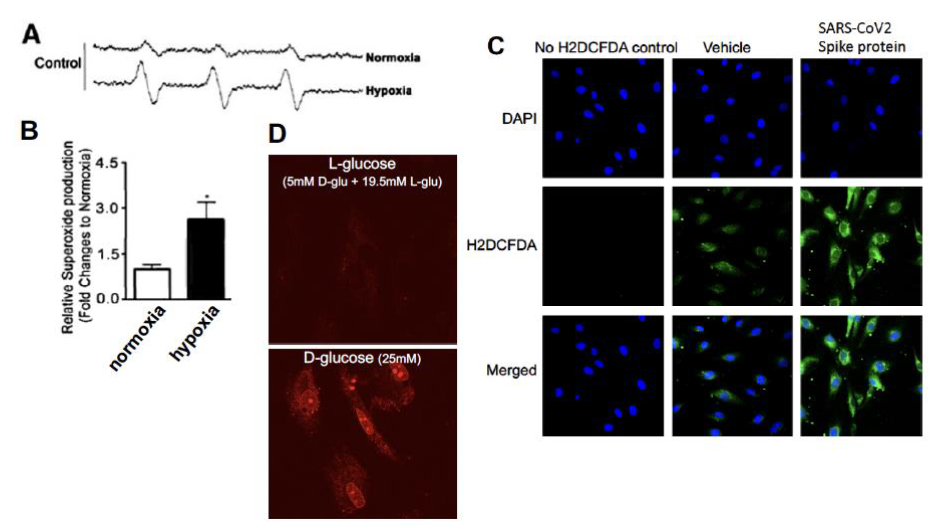
Figure C3. Measurement of reactive oxygen species in endothelial cells.
- EPR measurement of superoxide in ovine pulmonary endothelial cells under normoxia vs. hypoxia/reoxygenation.
- Relative superoxide. generation, as compared with normoxia of control cells in EPR.
- H2DCFDA flow cytometry of intracellular H₂O₂ in HLMVEC ± SARS-CoV-2 spike protein.
- MitoSOX Red assessment of mitochondrial ROS in human glomerular endothelial cells under high L- or D-glucose.
Core Leaders

Dr. Rudolf Lucas is Professor of Pharmacology in the VBC at MCG/AU, Dr. Lucas is a leading expert on proinflammatory mechanisms involved in endothelial dysfunction and barrier function, TNF and reactive oxygen species in diseases such as pneumonia, cerebral malaria, glomerulonephritis and obesity/type 2 diabetes as well as developing therapeutic targets. He has expertise in the isolation and culture of endothelial cells from numerous vascular beds.
Together with Co-Directors Drs. David Fulton and Masuko Ushio-Fukai, Dr. Lucas will serve as the Director of Core C.
Dr. David Fulton is Regents Professor and Director of the VBC at MCG/AU and a leading expert in the area of endothelial biology and NO/eNOS and NADPH oxidase in cardiovascular disease for the past twenty years.
Dr. Fulton will serve as a Co-I and provide additional studies and mechanistic insight on how glycolysis and PFKFB3 regulate eNOS function and redox balance. Dr. Fulton will also serve as co-director of Core C.
Core C Team

Rudolf Lucas
- Core C Lead

Dr. David Fulton
- Co-Investigator

Dr. Masuko Ushio-Fukai
- Contributor

Supriya Sridhar
- Research Associate

Maritza Romero Lucas
- Sr Research Scientist

Dipankar Ash
- Asst. Research Scientist

Stephen Haigh
- Postdoc Fellow
Vijay Patel
- Consultant

Richard McIndoe
- Contributor