- Augusta University
- Centers & Institutes
- Immunology Center of Georgia
- Adil Rasheed, PhD
Adil Rasheed, PhD
 Adil Rasheed's research focuses on the intersection of lipid metabolism with immune
cell fate & function in atherosclerotic cardiovascular disease.
Adil Rasheed's research focuses on the intersection of lipid metabolism with immune
cell fate & function in atherosclerotic cardiovascular disease.
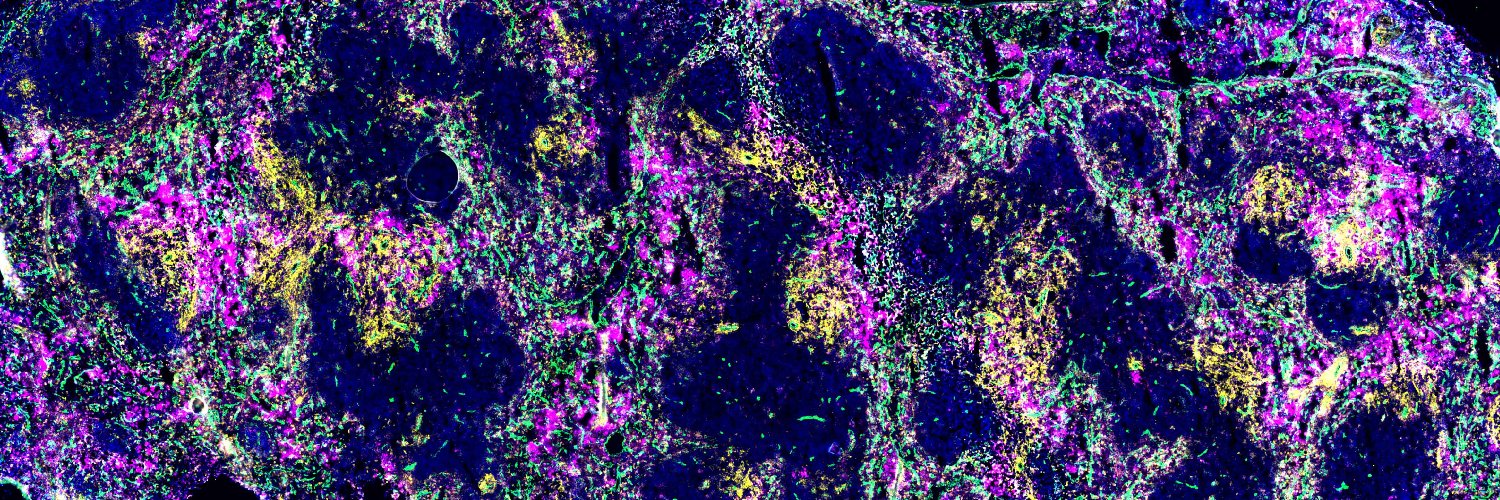
The atherosclerotic plaque develops over several decades, through the accumulation of excess circulating lipids and immune cells in the heart. One of these immune cells, macrophages, scavenge or “engulf” these lipids as a form of protection, leading to the foam cells that are characteristic of these atherosclerotic plaques. However, this process turns detrimental as lipid infiltration continues, leading to pathological features of atherosclerosis, such as increased inflammation and plaque remodeling, which ultimately progresses to the fatal complications of this disease, including heart attacks.
Rasheed’s research established a form of cell death, known as necroptosis, in the development of the necrotic core, which render these atherosclerotic plaques susceptible to rupture. The pseudokinase, the mixed lineage kinase domain-like protein (MLKL), was initially discovered in 2012 as the terminal regulator of the necroptosis signaling pathway. However, Rasheed’s work showed that at early stages, MLKL conversely has a protective role in limiting foam cell accumulation through promoting lipid mobilization in macrophages for metabolism via endocytosis.
But, not all foam cells are created equal. In fact, it has only recently been shown that in addition to macrophages, foam cells can also arise from the transdifferentiation of smooth muscle cells (SMC). While expressing macrophage markers, these SMC foam cells are deficient in several processes by comparison. Work from Rasheed and colleagues revealed that these SMC foam cells cannot efficiently engage autophagy to metabolize and remove excess lipids like macrophages, but that this can be rescued by treatment with metformin.
The immune cells that are recruited to the plaque during atherosclerosis are produced by a process known as hematopoiesis, where stem cells differentiate to these mature immune cells. This typically occurs in the bone marrow, but during inflammatory diseases the spleen also serves as a site of hematopoiesis. Through the study of MLKL, Rasheed’s work has demonstrated specific susceptibilities of the splenic niche, as compared to the bone marrow, to an environment of excess lipids.
Dr. Rasheed’s research will focus on the identification and targeting of molecular regulators of lipid metabolism as a therapeutic strategy for mediating immune cell contributions to atherosclerosis. These themes will also be used to explore immunometabolism in other complex diseases such as those of the metabolic syndrome, inflammatory diseases and cancer.
Understanding the molecular mechanisms underlying foam cell development in the atherosclerotic plaque
Assessing and mediating the influence of nutrient metabolism on immune cell phenotype
Identification of factors that influence the progression of atherosclerosis and advancement of the plaque
Investigate the adaptations of the hematopoietic niche in cardiovascular disease